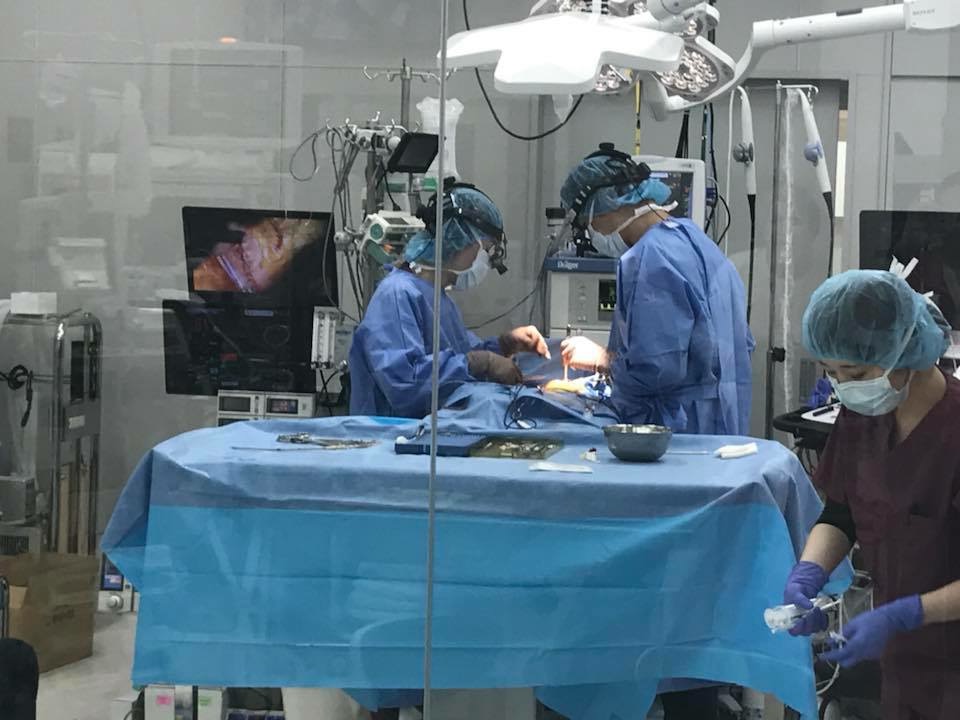
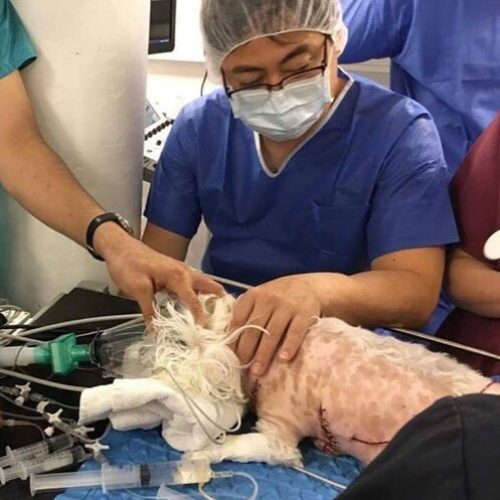
This is not a simple surgical procedure. It is a complete and very complex medical-surgical procedure from the preoperative phase to the hospital discharge 7-10 days later, where a team of 10 veterinarians intervene for a single dog.
Each veterinarian has a specific role, either medical or surgical. The procedure requires 2 surgeons (surgeon and one assistant surgeon), 3 sterile instrumentalists, 3 non-sterile assistants, 1 perfusionist, 1 anesthetist-resuscitator, and 1 cardiologist sonographer.
Here we will break down some of the processes of this procedure to give you a broad understanding of this complex intervention.
“The success of this intervention is due to the perfect harmony of a team that is perfectly in sync together where everyone has his or her designed roles, no technical details are left to chance.”
The “master machine” that makes open heart surgery possible is called the Cardio-Pulmonary Bypass (CPB) Machine.
In addition to this machine, the intervention requires 2 anesthetic machines – one with assisted ventilation (isoflurane or sevoflurane), a monitor to show live vital statistics, a thermal generator which cools or reheats the animal’s body on request at a precise temperature, a surgical table inclinable in all directions, an echograph with color doppler and a pediatric transesophageal probe, 4 syringe pumps and 2 infusion pumps, a coagulation time measuring device, a blood gas analyzer, a blood-biochemical analyzer, an electrolyte analyzer, an internal pediatric pallet defibrillator, a pace-maker, fresh blood compatible for transfusion (a machine called “cell-saver” in order to recycle the lost blood and carry out autotransfusions), a cardioplegic solution (which stops the heart), frozen sterile physiological serum, a large number of consumables and injectable medicines, conventional surgical aspirator equipment, a fine-tuned electric scalpel, microsurgical instruments, customized equipment (catheter, tubing, etc…) and more.
Extracorporeal Circulation
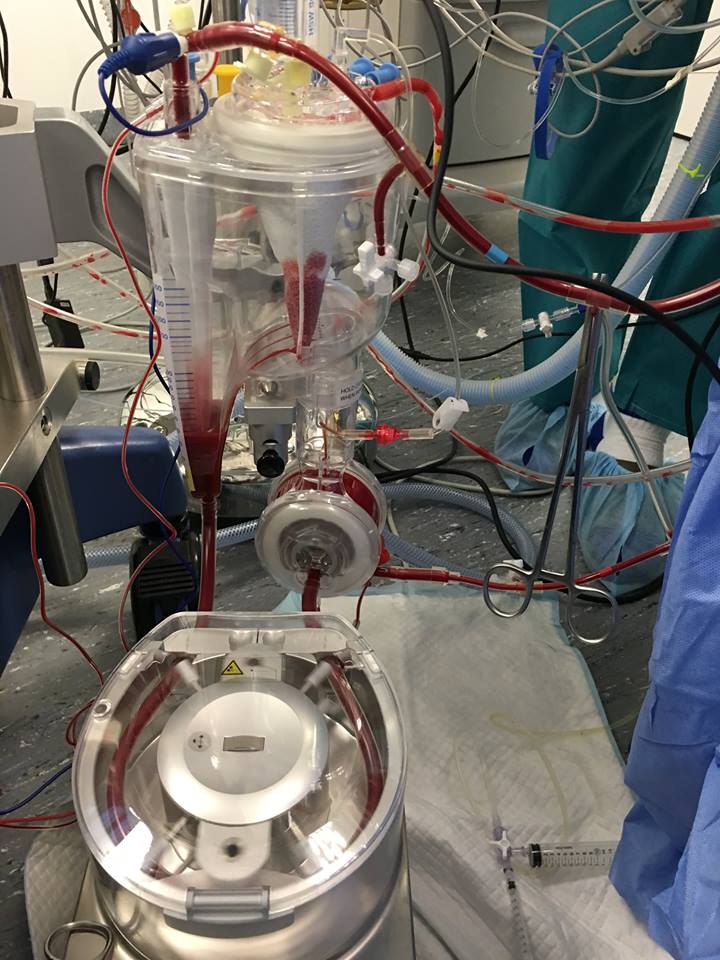
“This is one of the biggest constraints of the intervention for small breeds dogs. With special modifications They are able to operate on dogs weighing 2kg or less. Extracorporeal circulation techniques have been attempted in some medium or large sized dogs before without huge success.”
In dogs of any small size, difficulties are encountered given the low initial circulating blood volume.
Dr. Uechi has succeeded in developing an extracorporeal circulation procedure that does not use traditional techniques, and allows the operation of miniature dogs and even cats.
The use of extra-corporeal circulation can induce cardiac arrest and therefore work freely for the time required during the surgical open heart repair.
The CPB machine allows the heart and lungs to be taken over during surgery and requires: 3 pumps (one of which is equivalent to the heart pump), a system for mixing and exchanging blood cases (oxygenator, equivalent to the lungs), a system for controlling blood and body temperature (thermal generator), a tubing system adapted to the conformation of the dog.
The mastery of extracorporeal circulation by the perfusionist is crucial to the success of the intervention. Mastering extracorporeal circulation is the future in terms of treatment of heart disease in dogs.
Steps of Mitral Valve Repair Surgery
Prior to the induction of general anesthesia, the maximum number of steps is performed on animal vigil: insertion of 2 intravenous catheters (typically in anterior limbs), insertion of a urinary catheter (Foley catheter), shaving the left cervical area, the entire left chest wall, and the medial right posterior limb. The operating room is also prepared during this phase.
Premedication
General anesthesia
Surgical placement of an arterial catheter and venous catheter to measure invasive arterial blood pressure and central venous pressure throughout the procedure and especially during cardiotomy (heart stopping phase, approximately 2 hours).
Surgical opening of the rib cage, reclination of the lungs, pericardiectomy and access to the heart.
Dissection of the base of the aorta and minimal separation of the aorta and the pulmonary artery.
Administration of intravenous bolus heparin to induce a non-coagulable state. For this purpose, measurements of the activated partial thromboplastin time (ACT) are carried out until a minimum value of 300 seconds is obtained.
Laying of extracorporeal circulation cannulas. The left carotid artery and jugular vein are surgically isolated and a cannula is inserted into each vessel, their size depending on the type of cannulated vessel and the body weight of the dog.
Implementation of the partial and then total extracorporeal circulation. The cannulas are connected to the extracorporeal circulation circuit after carefully evacuating the air bubbles from the tubing. The arterial and venous lines are unclamped and the CPB starts at the adapted rate.
Transesophageal ultrasound: a pediatric transesophageal tube connected to an echocardiograph equipped with color flow doppler is inserted delicately in order to successfully obtain the median transverse and longitudinal views. The movement of the mitral leaflets and regurgitation are examined by the surgeon.
Catheterization of the ascending aorta (installation of the cardioplegia cannula): a catheter is gently inserted on the left lateral face of the ascending aorta.
Decreased body temperature: the body temperature is gradually decreased up to 28 degrees and maintained thanks to the thermal generator integrated in the extracorporeal circulation machine.
Injection of cardioplegic solution (via blood cardioplegia) in the coronary arteries via the aortic catheter and induction of cardiac arrest.
Incision of the left atrium and placement of the aspirations cannulas.
Pre-placement of artificial cordage’s: replacement of mitral tendon cords is the best technique for repairing broken cords. Double sutures of expanded polytetrafluoroethylene (e-PTFE or Gore-Tex) are placed between the papillary muscles (reinforced by pledgets to avoid tearing of the papillary muscle) and the mitral leaflets. Depending on the dog, a total of 4 to 6 new cords are laid.
Example pre-placement of artificial cordage’s:
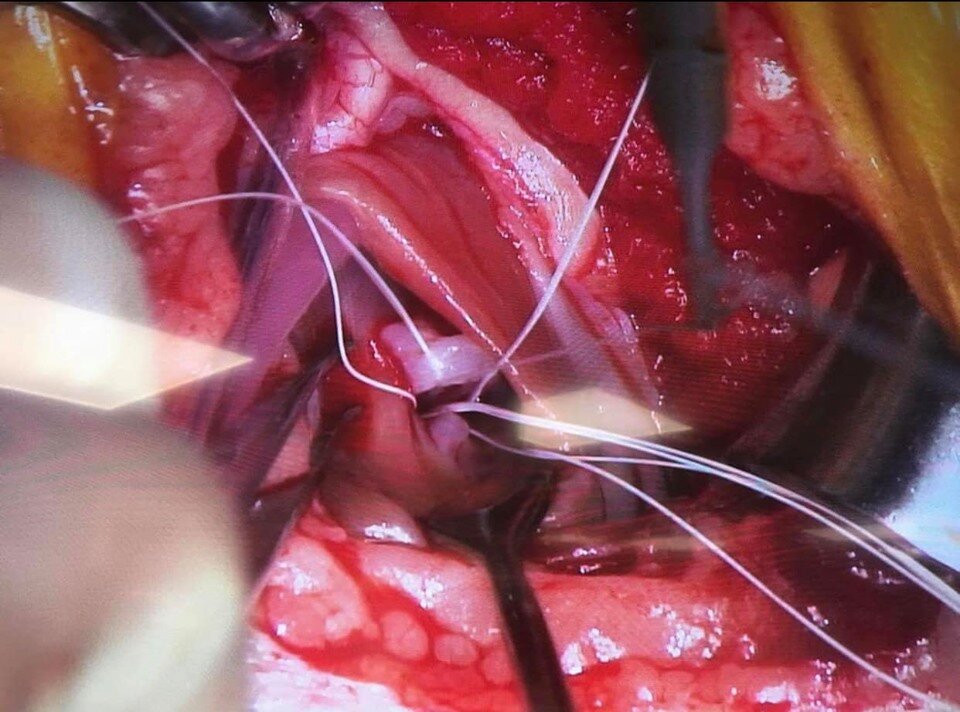
Mitral annuloplasty: tightens the mitral ring in order to decrease or even cancel the mitral regurgitation and thus to maintain a good systolic coaptation. The same material as the cords is used (ePTFE) in the form of two sutures reinforced by pledgets at the level of each commissure of the mitral ring.
Tightening of the replacement cords: the main challenge here is the difficulty in adjusting the cords, in length and tension, in order to obtain the optimum coaptation when closing the mitral valve. This phase is highly dependent on the surgeon’s experience and the mitral valve leaflets echostructure. The result is then tested with physiological serum under pressure in the left ventricular chamber by tilting the table.
Suture of the left atrium, evacuation of the intra-atrial air under transesophageal ultrasound control and declamping of the aorta.
Restarting the heartbeat: upon cessation of intracoronary administration of the cardioplegic solution, heartbeats are expected to resume spontaneously. A direct cardiac massage is performed systematically and if the heart does not restart on its own, a temporary pacemaker is placed or ventricular defibrillation is undertaken.
Transesophageal ultrasound recheck: the coaptation of the mitral leaflets and the mitral regurgitation are appreciated, which has usually decreased by at least 80%.
Progressive temperature rise in the body of the animal and gradual decrease in the flow rate of the arterial pump.
Removal of the carotid and jugular cannulas and termination of the CPB.
Continuous perfusion of protamine (heparin antagonist) over a 20-minute period. Simultaneous administration of continuous dobutamine infusion if invasive blood pressure is too low and unstable. Indeed, protamine can cause severe hypotension, bronchospasm and increased pulmonary vascular resistance. Ventilation and blood pressure should therefore be perfectly controlled at this time.
Closure of the thorax plane by plane (the pericardium is not sutured).
Chest tube placement.
Progressive waking over 2-3 hours: During this crucial phase, all the vital parameters and ventilation are closely controlled and their return to normal are obtained very gradually. Femoral catheters will be removed just before the actual wake-up (isoflurane stop) once invasive systolic blood pressure remains stable. The dog is then placed in an intensive care cage with oxygen supply and regulation of the ambient temperature as well as hygrometry.